Translating discoveries into benefits
Translational research is the process of translating basic scientific discoveries into medical interventions that improve public health.
NEI helps researchers, clinicians, and small businesses to overcome challenges in moving from scientific discovery to public health impact. These programs prevent promising medical interventions from being “lost in translation.”
Translational research helps bridge the gaps
This graphic shows the steps involved in converting a basic science discovery to a public health impact (top row) and indicates how translational research helps bridge the gaps between steps (bottom row).
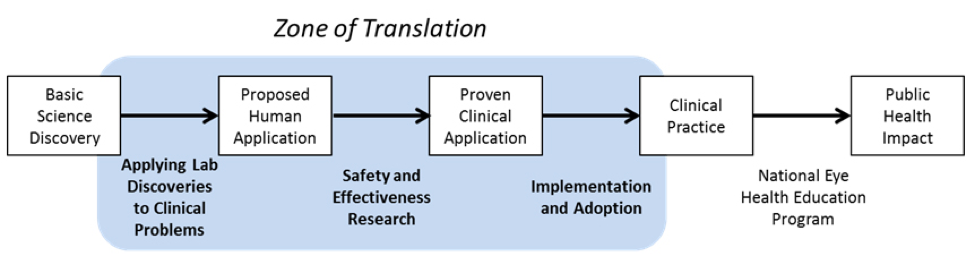
Based on Drolet and Lorenze, 2011
NEI supports translational research in many different ways.
Applying lab discoveries to clinical problems
While most NEI-funded studies investigate how the visual system works, NEI also supports laboratory and animal studies that explore treatments to prevent or reverse vision loss.
Safety and effectiveness research
NEI supports scientific studies that explore how new medicines, devices, or surgeries work in people. NEI also collaborates with the Food and Drug Administration (FDA) to facilitate the regulatory approval process for drugs and devices relating to eye health and eye diseases.
Implementation and adoption
NEI works with medical societies to encourage the adoption of proven interventions. NEI also conducts research about the comparative effectiveness of interventions to help doctors and patients make evidence-based decisions.
National Eye Health Education Program
The National Eye Health Education Program (NEHEP) helps health educators and community professionals increase awareness about eye health. NEHEP programs are designed to reach populations at higher risk for eye disease and vision loss and to promote the use of vision rehabilitation services.
Other NIH resources and funding opportunities
- NIH Resources for Translational Vision Research
- Collaborating with NEI Intramural Scientists
- National Center for Advancing Translational Sciences (NCATS)
- National Heart, Lung, and Blood Institute (NHLBI) Translational Research
- National Institute of Neurological Disorders and Stroke (NINDS) Translational Research
- Collaborating with Intramural Investigators at the NIH Clinical Center
To learn more
For more information about translational research at NEI, contact: