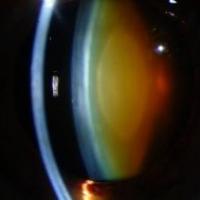
Current research
This section utilizes a number of different approaches to study inherited visual diseases affecting all parts of the eye.
In one approach to understanding inherited visual diseases we use positional cloning to identify genes important in human inherited diseases. Diseases currently undergoing linkage analysis, gene isolation, or characterization of mutations include retinitis pigmentosa, inherited cataracts, Bietti crystalline dystrophy, and several corneal dystrophies.
In a second approach, we attempt to establish associations between sequence changes in and around candidate genes and specific visual phenotypes. We have applied this type of study primarily to prevalent diseases with a complex inheritance pattern, including age related cataracts, myopia, and glaucoma.
Once a candidate gene has been identified and confirmed, the biochemical and pathophysiological implications of identified mutations are explored both in vitro through recombinant expression and functional characterization of wild type and mutant proteins and in vivo through transgenic expression or knock out of pathological proteins both in cultured cells and model organisms such as the mouse and zebrafish. One group of proteins we have investigated in detail are the lens crystallins, which make up more than 90 percent of the soluble protein of lens and are heavily modified in most cataracts. The effects that specific modifications produce on crystallin structure and other functions such as stability, resistance to damage by ultraviolet light, and formation of macromolecular aggregates are being studied. Examples of other molecules under study include genes implicated in retinal disease, such as CYP4V2, ZNF513, and KCNJ13.
Selected publications
Li, L., Chen, Y., Jiao, X., Jin, C., Jiang, D., Tanwar, M., Ma, Z., Huang, L., Ma, X., Sun, W., Chen, J., Ma, Y., M'Hamdi, O., Govindarajan, G., Cabrera, P.E., Li, J., Gupta, N., Naeem, M.A., Khan, S.N., Riazuddin, S., Akram, J., Ayyagari, R., Sieving, P.A., Riazuddin, S.A., Hejtmancik, J.F. Homozygosity Mapping and Genetic Analysis of Autosomal Recessive Retinal Dystrophies in 144 Consanguineous Pakistani Families. Invest Ophthalmol Vis Sci 2017;58:2218-2238.
Jiao, X., Li, A., Jin, Z.B., Wang, X., Iannaccone, A., Traboulsi, E.I., Gorin, M.B., Simonelli, F., Hejtmancik, J.F. Identification and population history of CYP4V2 mutations in patients with Bietti crystalline corneoretinal dystrophy. Eur J Hum Genet 2017.
Chen, J., Wang, Q., Cabrera, P.E., Zhong, Z., Sun, W., Jiao, X., Chen, Y., Govindarajan, G., Naeem, M.A., Khan, S.N., Ali, M.H., Assir, M.Z., Rahman, F.U., Qazi, Z.A., Riazuddin, S., Akram, J., Riazuddin, S.A., Hejtmancik, J.F. Molecular Genetic Analysis of Pakistani Families With Autosomal Recessive Congenital Cataracts by Homozygosity Screening. Invest Ophthalmol Vis Sci 2017;58:2207-2217.
Khan, S.Y., Vasanth, S., Kabir, F., Gottsch, J.D., Khan, A.O., Chaerkady, R., Lee, M.C., Leitch, C.C., Ma, Z., Laux, J., Villasmil, R., Khan, S.N., Riazuddin, S., Akram, J., Cole, R.N., Talbot, C.C., Pourmand, N., Zaghloul, N.A., Hejtmancik, J.F., Riazuddin, S.A. FOXE3 contributes to Peters anomaly through transcriptional regulation of an autophagy-associated protein termed DNAJB1. Nature communications 2016;7:10953.
Ma, X., Jiao, X., Ma, Z., Hejtmancik, J.F. Polymorphism rs7278468 is associated with Age-related cataract through decreasing transcriptional activity of the CRYAA promoter. Scientific reports 2016;6:23206.
Ma, Z., Yao, W., Chan, C.C., Kannabiran, C., Wawrousek, E., Hejtmancik, J.F. Human betaA3/A1-crystallin splicing mutation causes cataracts by activating the unfolded protein response and inducing apoptosis in differentiating lens fiber cells. Biochim Biophys Acta 2016;1862:1214-1227.
Chen, J., Ma, Z., Jiao, X., Fariss, R., Kantorow, W.L., Kantorow, M., Pras, E., Frydman, M., Riazuddin, S., Riazuddin, S.A., Hejtmancik, J.F. Mutations in FYCO1 Cause Autosomal-Recessive Congenital Cataracts. Am J Hum Genet 2011;88:827-838.
Riazuddin, S.A., Shahzadi, A., Zeitz, C., Ahmed, Z.M., Ayyagari, R., Chavali, V.R., Ponferrada, V.G., Audo, I., Michiels, C., Lancelot, M.E., Nasir, I.A., Zafar, A.U., Khan, S.N., Husnain, T., Jiao, X., Macdonald, I.M., Riazuddin, S., Sieving, P.A., Katsanis, N., Hejtmancik, J.F. A mutation in SLC24A1 implicated in autosomal-recessive congenital stationary night blindness. Am J Hum Genet 2010;87:523-531.
Li, L., Nakaya, N., Chavali, V.R., Ma, Z., Jiao, X., Sieving, P.A., Riazuddin, S., Tomarev, S.I., Ayyagari, R., Riazuddin, S.A., Hejtmancik, J.F. A mutation in ZNF513, a putative regulator of photoreceptor development, causes autosomal-recessive retinitis pigmentosa. Am J Hum Genet 2010;87:400-409.
Ma, Z., Piszczek, G., Wingfield, P.T., Sergeev, Y.V., Hejtmancik, J.F. The G18V CRYGS Mutation Associated with Human Cataracts Increases γS-Crystallin Sensitivity to Thermal and Chemical Stress. Biochemistry 2009;48:7334-7341.
Jiao, X., Yang, Z., Yang, X., Chen, Y., Tong, Z., Zhao, C., Zeng, J., Chen, H., Gibbs, D., Sun, X., Li, B., Wakins, W.S., Meyer, C., Wang, X., Kasuga, D., Bedell, M., Pearson, E., Weinreb, R.N., Leske, M.C., Hennis, A., Dewan, A., Nemesure, B., Jorde, L.B., Hoh, J., Hejtmancik, J.F., Zhang, K. Common variants on chromosome 2 and risk of primary open-angle glaucoma in the Afro-Caribbean population of Barbados. Proc Natl Acad Sci USA 2009;106:17105-17110.
Ophthalmic Molecular Genetics Section key staff
| Name | Title | Phone | |
|---|---|---|---|
| Rongfang Chen | Visiting Fellow | rongfang.chen@nih.gov | |
| Rebecca Clark | Postbaccalaureate Fellow | rebecca.clark2@nih.gov | 301-496-3577 |
| J. Fielding Hejtmancik, M.D., Ph.D. | Senior Investigator | hejtmancikj@nei.nih.gov | 301-496-8300 |
| Huan Hu | Visiting Fellow | huan.hu@nih.gov | 301-496-6999 |
| Xiaodong Jiao, M.D. | Biologist | xj3w@nih.gov | 301-435-2586 |
| Zhiwei Ma, M.D., Ph.D. | Staff Scientist | zhiwei.ma@nih.gov | 301-594-7345 |
| Yan Ma | Research Associate | yan.ma@nih.gov | 301-435-2585 |
| Hang Qi | Visiting Fellow | hang.qi@nih.gov | 301-435-1270 |
| Mohd Hussain Shah, Ph.D. | Visiting Fellow | mohd.shah@nih.gov | |
| Gaohui Zhou | Visiting Fellow | gaohui.zhou@nih.gov |