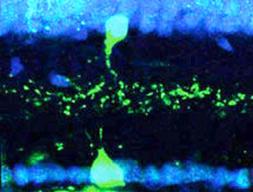
A VG3 amacrine cell (top, in green) is shown superimposed with an object motion detector cell (bottom, green). Each of the neurons is expressing sidekick-2, an adhesion molecule that allows the two neurons to find each other and connect.
When we move our head, the whole visual world moves across our eyes. Yet we can still make out a bee buzzing by or a hawk flying overhead, thanks to unique cells in the eye called object motion sensors. A new study on mice helps explain how these cells do their job, and may bring scientists closer to understanding how complex circuits are formed throughout the nervous system. The study was funded by the National Institutes of Health, and was published online in Nature.
“Understanding how neurons are wired together to form circuits in the eye is fundamental for advancing potential new therapies for blinding eye diseases,” said Paul A. Sieving, M.D., Ph.D., director of NIH’s National Eye Institute (NEI). “Research aimed at regenerating photoreceptors, for example, is enriched by efforts to understand the circuitry in the eye.”
Object motion sensors are one of about 30 different types of retinal ganglion cells (RGCs) in the retina, the light-sensitive tissue at the back of the eye. These cells are unique because they fire only when the timing of a small object’s movement differs from that of the background; they are silent when the object and the background move in sync. Researchers believe this is critical to our ability to see small objects moving against a backdrop of complex motion.
The cells in the retina are wired up like an electrical circuit. Vision begins with photoreceptors, cells that detect light entering the eye and convert it into electrical signals. Middleman neurons, called interneurons, shuttle signals from photoreceptors to the RGCs. And each RGC sends the output visual information deeper into the brain for processing. This all takes place within fractions of a second, so the scientists were surprised to discover that before it reaches object motion sensors, visual information about object motion takes a detour through a unique type of interneuron. Their results represent an ongoing effort by scientists to map out complex circuits of the nervous system.
“Getting these connections precisely correct is incredibly important, as each specific feature of vision, such as seeing a particular direction of motion or a color, relies on it,” said the study’s lead investigator, Arjun Krishnaswamy, Ph.D., of Harvard University’s Center for Brain Science, Cambridge, Massachusetts. “It’s also incredibly complex. Within the retina, all these different types of RGCs and interneurons intermingle as they develop. There have to be remarkable ways to sort them out so they connect up properly.”
Using a genetically engineered mouse line, the researchers recorded the activity of object motion sensors and found that the cells form synapses (or connections) with interneurons called VG3 amacrine cells. What’s interesting about this connection is that most retinal circuits tend to follow a more direct, and therefore faster, route. RCGs typically are two synapses away from a photoreceptor, but with the addition of VG3 amacrine cells to the circuit, object motion sensors appear to be three synapses away, slowing visual information delivered to the cells.
To test this idea, the scientists flashed light on the retinas of the mice and found that on average the object motion sensors responded later than other types of retinal ganglion cells. They also selectively activated the sensors by projecting light patterns onto the retinas that mimicked the movement of small objects against a desynchronized background. Mice with genetically eliminated VG3 amacrine cells did not show these responses.
The researchers theorize that the longer pathway contributes to an essential delay, ensuring that information from the central field of view and from the periphery arrive at the object motion sensor at the same time. This in turn allows the object motion sensors to accurately assess the difference between the motion of a hawk and the slow-moving clouds above it, or the flight of a baseball and the undulating crowd in the stadium.
Dr. Krishnaswamy and his colleagues then investigated how the circuit develops. They found that object motion sensors and VG3 amacrine cells each make a protein called sidekick-2, specifically where they contact each other. Sidekick-2 is an adhesion molecule that allows the two cell types to find each other and stick together so they can communicate across a synapse.
Mice genetically engineered to block sidekick-2 production lacked synapses connecting VG3 cells to object motion sensors. Moreover, electrical recordings showed that sensors in these mice did not distinguish the motion of a small object from background motion.
For Dr. Krishnaswamy, the next steps are to investigate the role of sidekick-2 in brain development, first in mice and eventually in humans.
“Neurons in the brain work just like neurons in the retina, and scientists are beginning to understand how they make precise connections to form circuits that control thoughts, senses and emotions,” said Edmund Talley, Ph.D., program director at the National Institute of Neurological Disorders and Stroke (NINDS), part of the NIH. “This pioneering work demonstrates the molecular specificity behind these connections.”
The study was conducted in the lab of Joshua R. Sanes, Ph.D. Dr. Sanes serves on the steering committee for the NEI Audacious Goals Initiative (https://nei.nih.gov/audacious) on neuro-regenerative medicine. The aim is to restore vision by regenerating neurons and neural connections in the eye and visual system.
This research was supported in part by NEI grant EY022073 and NINDS grant NS029169
Reference:
A. Krishnaswamy, et al. “Sidekick2 directs formation of a retinal circuit that detects differential motion,” Nature, August 2015. doi:10.1038/nature14682
Contacts:
Kathryn DeMott, Joe Balintfy
NEI
(301) 496-5248
neinews@nei.nih.gov
Christopher G. Thomas
NINDS
nindspressteam@ninds.nih.gov
(301) 496-5751