This year, the Food and Drug Administration (FDA) approved Syfovre (pegcetacoplan) and Izervay (avacincaptad pegol), the very first drugs for treating geographic atrophy (GA), also known as late-stage “dry” age-related macular degeneration (AMD). Both drugs target the immune system’s complement pathway, and while the drugs were brought to market by the private sector, they represent years of support from the National Eye Institute (NEI) in the form of grants to research campuses around the country.
Syfovre (pegcetacoplan) and Izervay (avacincaptad pegol) are approved to treat geographic atrophy, also known as late-stage "dry" age-related macular degeneration. They are administered through eye injection. Adobe Stock.
“This is truly an exciting time for the field and underscores the importance of basic and translational scientific research,” said NEI Director Michael F. Chiang, M.D. “These advances in AMD therapeutics wouldn’t have happened without federal support through funding from the National Eye Institute.”
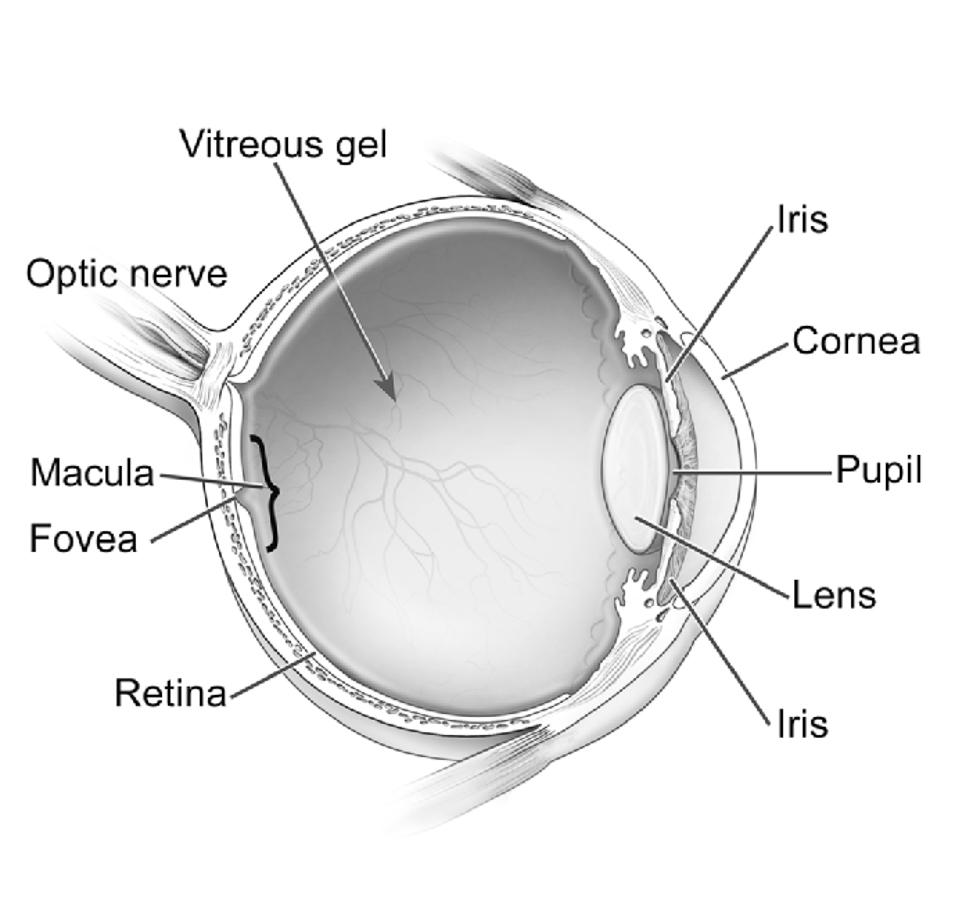
Cross-section of the eye. National Eye Institute.
AMD is the leading cause of vision loss and blindness for Americans aged 65 years and older. An estimated 20 million Americans have AMD. Of these, about 1.5 million have vision-threatening AMD.1 AMD affects the light-sensing tissue in the back of the eye called the retina. Most vision loss occurs when AMD damages the macula, a central part of the retina responsible for sharp, straight-ahead vision.

Caption: Simulated vision loss from advanced AMD. National Eye Institute.
The AMD-complement connection
The complement pathway protects against pathogens by triggering a cascade of proteins that enhance the body’s immune response. Complement proteins C1 through C9 provide the “gas” for activation, while others like complement factor H (CFH) and complement factor I (CFI) act as “brakes.” Working together, these opposing pedals enable the body to fight infection and destroy damaged cells while sparing healthy tissue. But slight variations in complement proteins can upset this balance, leading to disease states.
In 2005, an NEI-funded research team discovered strong associations between CFH and AMD as part of the first genome-wide association study (GWAS).2 GWASs enable comparisons of whole genomes (the complete set of human genes) of hundreds or thousands of people to isolate associations between traits such as increased disease risk and genetic variants. Four other NEI-funded studies that published at around the same time also associated the same CFH variant with AMD risk.3-6 The results led scientists to hypothesize that AMD was part of an inflammatory response, possibly triggered by other known AMD risk factors such as diet and smoking. In light of these findings, NEI assembled the International AMD Genomics Consortium, comprising 33 research institutions. The effort identified 34 regions of the human genome associated with AMD that could potentially serve as drug targets.7
Dry versus wet AMD
Late-stage AMD comes in a wet form, so-called because abnormal new blood vessels leak fluid into the macula, potentially causing vision loss. Wet AMD accounts for about 10 percent of AMD cases. GA is the late stage of the dry form of AMD, which is defined by retinal lesions. Laser therapies became available in 1982 for wet AMD. In the early 2000s, ophthalmologists began treating wet AMD by blocking abnormal blood vessel activity in the retina by injecting anti-vascular endothelial growth factor (VEGF) into the eye. Anti-VEGF drugs such as Avastin (bevacizumab) and Lucentis (ranibizumab) not only slowed wet AMD progression but, in most cases, reversed vision loss—a major game-changer for patients with wet AMD.
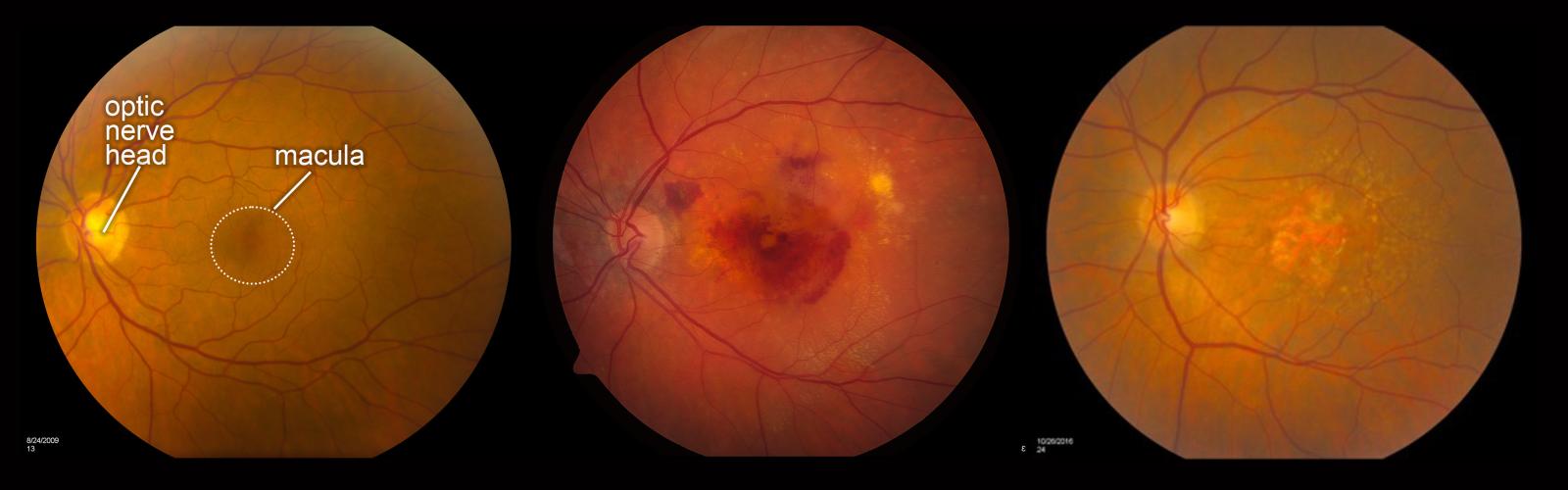
Photos of the retina. From left to right are a normal retina, a retina with wet AMD (neovascularization), and a retina with dry AMD (geographic atrophy). Emily Chew, National Eye Institute.
The watershed discovery of CFH’s role in AMD—and subsequent associations of other CFH region variants and with other complement genes—opened the door for targeting complement proteins with drug therapies; however, translating these discoveries into therapies for dry AMD was slower.
Homing in
In 2013, three studies, led by Anand Swaroop, Ph.D., NEI Intramural Research Program; NEI grantee Johanna Seddon, Ph.D., formerly of Tufts Medical Center; and a commercial team at deCode Genetics in Iceland, converged on a rare variant of the C3 gene that increases AMD risk by about 3-fold. The finding suggested that suppressing the complement system, and particularly C3, could be useful against AMD.8-10 Homing in on control of the eye’s immune system had finally paid off.
C3 plays a role in each of three parallel complement cascades, known as the classical, alternative, and lectin pathways. NEI funded complement expert John Lambris, Ph.D., University of Pennsylvania, to study C3’s role in AMD and to develop methods to activate or inhibit it. Lambris filed several patents for compstatins, molecules that modify C3 function. His discoveries led to the development of Syfovre for GA by Apellis Pharmaceuticals.
Izervay inhibits complement factor 5 protein (C5). C5 plays a different role in the complement system, operating downstream of C3 at the point where the three complement cascades converge. C5 is elevated in AMD patients, and inhibiting this molecule may preserve specific anti-inflammatory properties of C3. Research into the role of C5 in AMD also received public funding, from both NIH and the Veteran’s Administration.
Clinical trials
In clinical trials, both Syfovre and Izervay demonstrated an ability to slow the rate of GA progression, preserving sharp, central vision. Monthly administration of Syfovre decreased the rate of lesion growth by 18-22% over two years.11 Monthly administration of Izervay decreased the rate of lesion growth by 14% over one year.12
“Small effects of these drugs on GA are not surprising, as these molecules are targeted to one complement component,” noted Swaroop. “GA is a complex trait and not a single gene disease, he added.
But while both Syfovre and Izervay slow GA progression, patients receiving either drug are at an increased risk for developing wet AMD. Additionally, since FDA approval, the administration of Syfovre has been associated with a very rare but potentially severe inflammatory reaction called occlusive retinal vasculitis. While therapies to treat wet AMD have become increasingly effective, some physicians and patients may hesitate to treat their GA with Syfovre or Izervay for concerns about developing wet AMD or other complications.
The future
Results of follow-up studies of Syfovre and Izervay will be important to fully understand the long-term impacts and, importantly, the risks of these new therapies, said Emily Chew, M.D., also of the NEI Intramural Research Program. “Does the treatment effect continue, accelerate, or does the effect level off? If there is an increase or a maintenance of the treatment effect, one would have greater confidence in the therapy.”
Follow-up research is also needed to understand which patients benefit the most from these new treatments, relative to the risks. The impact of GA on vision depends on where the damage is located in the eye—the closer to the macula, the greater the visual deficit, which may influence the decision to treat.
“We’re at the very early stages,” said Rajendra Apte, M.D., Ph.D., Washington University in St. Louis, who was not involved in the development of these drugs. “We have our first therapeutics for GA, but it’s like photodynamic laser therapy for wet AMD, our goal was to reduce the risk of severe vision loss in a subset of patients.”
The approvals of Syfovre and Izervay represent the culmination of years of NEI-sponsored research and a launching point for future innovation. In addition to providing the first treatment option for dry AMD patients, the success of these new drugs provides proof-of-concept to target complement. Researchers are exploring additional drugs that target the complement cascade.
References
- Rein DB, Wittenborn JS, Burke-Conte Z, et al. Prevalence of Age-Related Macular Degeneration in the US in 2019. JAMA Ophthalmology. 2022;140(12):1202-1208. doi:10.1001/jamaophthalmol.2022.4401
- Klein RJ, Zeiss C, Chew EY, et al. Complement factor H polymorphism in age-related macular degeneration. Science. Apr 15 2005;308(5720):385-9. doi:10.1126/science.1109557
- Edwards AO, Ritter R, 3rd, Abel KJ, Manning A, Panhuysen C, Farrer LA. Complement factor H polymorphism and age-related macular degeneration. Science. Apr 15 2005;308(5720):421-4. doi:10.1126/science.1110189
- Hageman GS, Anderson DH, Johnson LV, et al. A common haplotype in the complement regulatory gene factor H (HF1/CFH) predisposes individuals to age-related macular degeneration. Proceedings of the National Academy of Sciences of the United States of America. May 17, 2005 2005;102(20):7227-7232. doi:10.1073/pnas.0501536102
- Haines JL, Hauser MA, Schmidt S, et al. Complement factor H variant increases the risk of age-related macular degeneration. Science. Apr 15 2005;308(5720):419-21. doi:10.1126/science.1110359
- Zareparsi S, Branham KE, Li M, et al. Strong association of the Y402H variant in complement factor H at 1q32 with susceptibility to age-related macular degeneration. Am J Hum Genet. Jul 2005;77(1):149-53. doi:10.1086/431426
- Fritsche LG, Igl W, Bailey JN, et al. A large genome-wide association study of age-related macular degeneration highlights contributions of rare and common variants. Nat Genet. Feb 2016;48(2):134-43. doi:10.1038/ng.3448
- Helgason H, Sulem P, Duvvari MR, et al. A rare nonsynonymous sequence variant in C3 is associated with high risk of age-related macular degeneration. Nat Genet. Nov 2013;45(11):1371-4. doi:10.1038/ng.2740
- Seddon JM, Yu Y, Miller EC, et al. Rare variants in CFI, C3 and C9 are associated with high risk of advanced age-related macular degeneration. Nat Genet. Nov 2013;45(11):1366-70. doi:10.1038/ng.2741
- Zhan X, Larson DE, Wang C, et al. Identification of a rare coding variant in complement 3 associated with age-related macular degeneration. Nat Genet. Nov 2013;45(11):1375-9. doi:10.1038/ng.2758
- Heier JS, Lad EM, Holz FG, et al. Pegcetacoplan for the treatment of geographic atrophy secondary to age-related macular degeneration (OAKS and DERBY): two multicentre, randomised, double-masked, sham-controlled, phase 3 trials. Lancet. Oct 21 2023;402(10411):1434-1448. doi:10.1016/s0140-6736(23)01520-9
- Khanani AM, Patel SS, Staurenghi G, et al. Efficacy and safety of avacincaptad pegol in patients with geographic atrophy (GATHER2): 12-month results from a randomised, double-masked, phase 3 trial. Lancet. Oct 21 2023;402(10411):1449-1458. doi:10.1016/s0140-6736(23)01583-0